The Canterbury Psyllid Liberibacter Initiative can be read about in full here https://potatoesnz.co.nz/rd-project/canterbury-psyllid-liberibacter-initiative/ The CPLI Degree Day Graphs are one of the activities from this project.
These following graphs show accumulated degree days (DD) for Pukekohe, Hawke’s Bay, Manawatu, Mid-Canterbury and South Canterbury from 2014-2020. The graph for each region also contains accumulated degree days for a hot and cold year in that region, to allow you to make comparisons with the current season. The number of psyllid (TPP) generations for the current season so far is indicated on the graph with a yellow arrow.
| Name | Downloads |
|---|---|
| Name | Downloads |
|
summary_table_10_Feb_2020
|
489 downloads |
About Degree Days
Insecticides that are applied for a perennial insect pest based on a calendar date often result in poor insect control and a waste of resources. Insect activity varies from year to year depending on weather.
As long as accurate weather data can be obtained, using degree days to time treatments is more reliable than a calendar date and allows growers to pinpoint a specific treatment date each year.
Degree days (often referred to as “growing degree days”) are accurate because insects have a predictable development pattern based on heat accumulation.
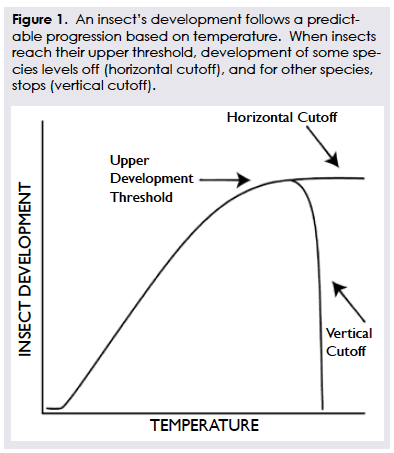
Every insect requires a consistent amount of heat accumulation to reach certain life stages, such as egg hatch or adult flight. Degree day values interpret that heat accumulation. When used to determine treatment timing, they are an important component of an Integrated Pest Management program, providing a cost-effective tool to reduce insect feeding damage (See Figure 1).
Things to consider:
Psyllids are active throughout the year and therefore 1 July is an arbitrary starting point.
It is important to be aware of other plants near your crop that can sustain TPP and act as a source of infestation. These include nightshade weeds, African boxthorn, poroporo but also volunteer potatoes.
* Since the psyllid research is a work in progress, we strongly advise you to use the degree day graphs in conjunction with crop monitoring using sticky traps and plant sampling. Crop monitoring provides valuable information on TPP arrival, population build up and the life stages present in your crop (see the links below for a sticky trap monitoring brochure).
What do you need to know about insects and degree days?
Insect growth and development
Since insects are cold-blooded, temperature plays a major role in their growth and development. Insects require a certain amount of heat to develop from one stage in their life to another i.e. eggs – nymphs – adults. Insects have a lower development threshold below which insect development is negligible and an upper development threshold which is the maximum temperature before development stops. These thresholds can be used in predicting insect development.
Further Information
Using Degree Days to Time Treatments for Insect Pests by Marion S. Murray, Utah State University.
Understanding Degree Days by Gabriella Zilahi-Balogh and Douglas G. Pfeiffer, Virginia Polytechnic Institute and State University.
Spraying: Unfortunately, the potato psyllid is a difficult pest to control. Spraying can be reasonably effective but it’s essential to cover all parts of the plant thoroughly with spray. This can be difficult because potato plants have dense foliage later in the growing season. If you do spray, remember to follow all aspects of Good Agricultural Practice, including spraying guidelines ensuring you don’t spray too close to harvesting time. Also, make sure you follow resistance management guidelines to prevent psyllid becoming resistant to the sprays.
Weeds: Another control is to keep down or eradicate other plants that are alternative hosts to psyllid. These include common weeds such as convolvulus, jimson weed (thornapple), mallow, African boxthorn (Lycium ferocissimum and black nightshade. Ornamental solanaceous plants are also potential hosts, and so is the native plant poroporo. Removing these plants from around your potatoes may reduce the likelihood of psyllid.
Contact
For further information please contact Natasha Taylor, Research Associate, Plant and Food Research, ph: (06) 975 8880



